A Review of Some Approximate Methods Used in Aerodynamic Heating Analyses
Abstract
Background
In aerodynamic levitation, solids and liquids are floated in a vertical gas stream. In combination with CO2-laser heating, containerless melting at high temperature of oxides and silicates is possible. We apply aerodynamic levitation to bulk rocks in training for microchemical analyses, and for evaporation and reduction experiments.
Results
Liquid silicate droplets (~2 mm) were maintained stable in levitation using a nozzle with a 0.8 mm bore and an opening bending of lx°. The gas menstruation was ~250 ml min-1. Stone powders were melted and homogenized for microchemcial analyses. Light amplification by stimulated emission of radiation melting produced chemically homogeneous glass spheres. Simply highly (eastward.m. H2O) and moderately volatile components (Na, One thousand) were partially lost. The limerick of evaporated materials was determined by straight combining levitation and inductively coupled plasma mass spectrometry. It is shown that the evaporated material is composed of Na > K >> Si. Levitation of metal oxide-rich material in a mixture of H2 and Ar resulted in the exsolution of liquid metal.
Conclusions
Levitation melting is a rapid technique or for the preparation of bulk rock powders for major, small-scale and trace chemical element assay. With exception of moderately volatile elements Na and K, bulk rock analyses can be performed with an uncertainty of ± 5% relative. The technique has nifty potential for the quantitative determination of evaporated materials from silicate melts. Reduction of oxides to metal is a means for the extraction and analysis of siderophile elements from silicates and tin can be used to better understand the origin of chondritic metal.
Background
Aerodynamic levitation
The term aerodynamic levitation is used for a technique, in which solids or liquids are freely floated on height of a vertical gas stream. With this technique, samples are non in contact with any container textile. Therefore, aerodynamic levitation allows the conduction of high-temperature experiments while avoiding bug related to the chemical interaction between sample and container walls (e.g., corrosion of oxide crucibles by silicate melts [1, two], proceeds or loss of siderophile elements and Fe in Pt crucibles, [3, four]).
Oxides and silicates tin be heated and melted with a COtwo gas laser [5], a mirror furnace [six], or a solar furnace [vii], while they are floating on height of the gas stream. Depending on the supplied energy, temperatures >3000°C tin can be reached [8].
Aerodynamic levitation has been used for studies of the physical properties of solids [5] and liquids [9–thirteen] at high temperatures. It has as well been used to investigate the crystallization beliefs of oxides [14] and silicates [xv–17]. Due to the absence of heterogeneous nucleation sites, aerodynamic levitation was too used for the grooming of glasses from substances that otherwise crystallize during cooling [18–20].
In this contribution, we describe an aerodynamic levitation device and demonstrate advantages and limitations of aerodynamic levitation in combination with laser heating for a) sample training for bulk rock chemical analyses, b) high-temperature evaporation experiments and c) reduction experiments.
Application fields of aerodynamic levitation in Geosciences
Sample training for bulk rock chemical analyses
Bulk rock major, minor and trace chemical element analyses are an integral part of modern geological studies [21]. Conventionally, bulk rock analyses are obtained by X-ray fluorescence on fused glass disks, by mass or by optical spectroscopy of dissolved materials, or by instrumental neutron activation assay. Recently, major, pocket-sized and trace element concentrations take been adamant on fused bulk rock samples by laser ablation inductively coupled mass spectrometry (LA-ICPMS) [22–26]. Bulk rock spectacles were prepared past means of fusion with Li-borate flux in a Pt crucible [22, 25] or by melting on an electrically heated Ir strip [23, 27]. The latter technique requires less material ( < 50 mg), but results in the loss of some volatile elements from the sample.
Pack et al. [28], Pack [29] and Patzer et al. [thirty] used aerodynamic levitation in conjunction with COtwo light amplification by stimulated emission of radiation melting as grooming technique for analyses of Ca, Y, REEs, Zr, and Hf in bulk chondrites and achondrites. The analyses were conducted using LA-ICPMS. The concentration of Ca, which was used as internal standard, was determined by electron microprobe analyses (EPMA). Simply about ten mg of sample material was required. In Pack et al. [28], 17 chondrites were analyzed for Y and Ho at concentration levels of ~2.4 μg g-1 and ~0.09 μg k-ane. Small variations ( ± 5%) of the Y/Ho ratios were related to fractional condensation processes in the solar nebula. Pack [29] demonstrated that REEs can be determined with a precision better than ± v% on fused majority meteorites. It was demonstrated that majority chondrites do not have unfractionated REE abundances. Patzer et al. [30] showed that precise and authentic bulk rock Zr/Hf ratios tin be obtained by levitation melting and subsequent LA-ICPMS analysis. Fractionations among refractory lithophile elements were related to processes in the solar nebula 4.half dozen Ga agone.
Details of the levitation appliance, yet, were not described past Pack et al. [28], Pack [29] and Patzer et al. [thirty] and will exist presented in this contribution. It volition likewise be demonstrated that laser-assisted melting of ~10 mg rock powders is suitable not only for refractory trace element (Y, REE, Zr, Hf), but besides for bulk major and modest element analyses.
Evaporation experiments
Brine evaporation during chondrule melting
Alkalis belong to the group of moderately volatile elements [31]. In cosmochemical context, evaporation of alkalis provides important insights into the conditions (pressure, oxygen fugacity, duration) of chondrule melting in the protoplanetary disk [32, 33]. Chondrules are ~0.1 - 1 mm sized silicate spheres (Fig. i) that formed by a cursory, but intense melting consequence in the solar nebula [34]. They are major constituents of chondritic meteorites. In a low-pressure (10-half dozen to 10-4 bar), H2-rich nebular gas, alkalis are expected to evaporate within minutes from the melt. However, it was shown past Borisov et al. [33] and Alexander et al. [32] that chondrules did not lose alkalis during melting. Melting of chondrules in a non-canonical high-pressure nebular gas [35] may explain the observed absence of alkali evaporation.

Back scattered electron image of a chondrule from the Renazzo (CR2) carbonacenous chondrite [modified later on [37]]. The chondrule contains olivine, metal and intersitial mesostasis.
We will demonstrate that evaporation of alkalis from levitated molten silicates can exist monitored with loftier time resolution. The applicability of the experimental procedure with respect to the problem of alkali-retention in chondrules volition be discussed.
In-situ reduction of silicate melts
Chondrites and chondrules incorporate metal that did not form by direct condensation from the solar nebula. Instead, it has been suggested that metal in chondrites formed by reduction from oxides during the chondrule melting issue [36]. Some chondrules contain Iron, Ni metal blebs that may have formed by reduction during the cursory chondrule melting event (Fig. 1, modified after [37]). In this contribution, we demonstrate that Fe, Co, Ni and likely other and more than siderophile elements can be transferred from the oxidized course into liquid metal during levitation.
The usability of the technique for the analysis of siderophile trace elements in rocks will also be discussed.
Results
The aerodynamic levitation apparatus and sample preparation
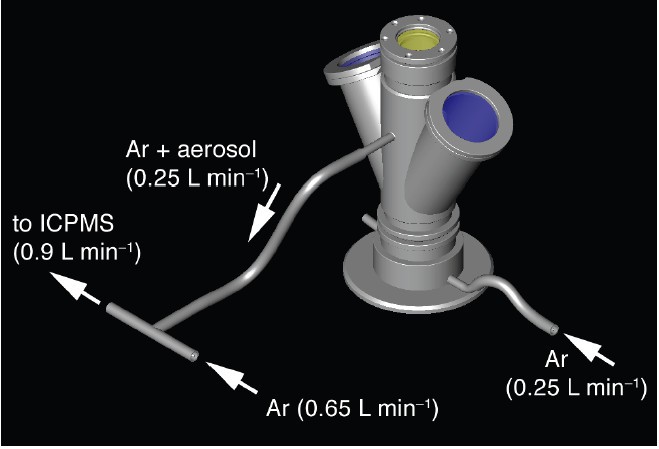
The levitation device comprised an infrared (IR) CO2 laser heat source, a levitation bedroom with levitation nozzle and a levitation gas supply (Fig. 2).
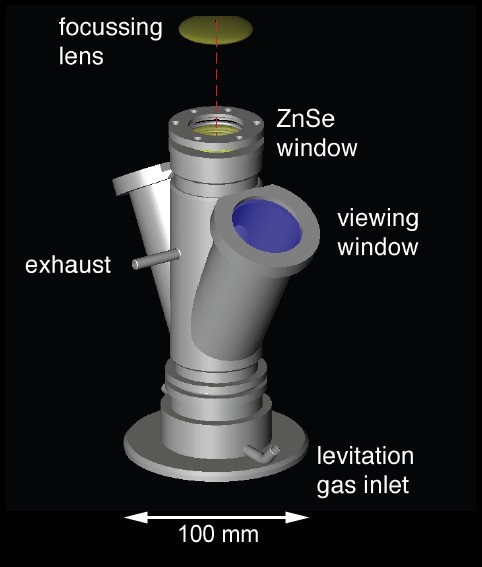
Sketch illustrating the levitation apparatus. The levitation nozzle was placed inside the sample chamber. The chamber comprised sapphire windows for viewing and illumination and a ZnSe window for the laser beam. The levitation gas could be conducted via the exhaust to the source of an ICPMS for chemical analyses.
Nosotros used a SYNRAD l Westward CO2 light amplification by stimulated emission of radiation (λ = 10.4 μm) as rut source. The laser was focused to a beam bore of ~1.5 - 2 mm by means of a ZnSe lens (ƒ = 125 mm). The laser free energy output could continuously be varied between 0 and 95%. The levitation sleeping room hosted the levitation nozzle. The chamber was used when conducting experiments in controlled atmospheres (e.grand., under reducing conditions) were carried out (or the chamber was used during conducting experiments in controlled atmospheres). Two sapphire windows were used for video monitoring and for illumination. The tiptop of the bedroom was covered with an IR transparent ZnSe window. The levitation nozzle had an opening angle of threescore° and a 0.8 mm bore (Fig. 3). It was made of aluminum. The levitation gas was regulated with a computer-controlled VÖGTLIN red-y mass flow controller (0 - k mL min-1).
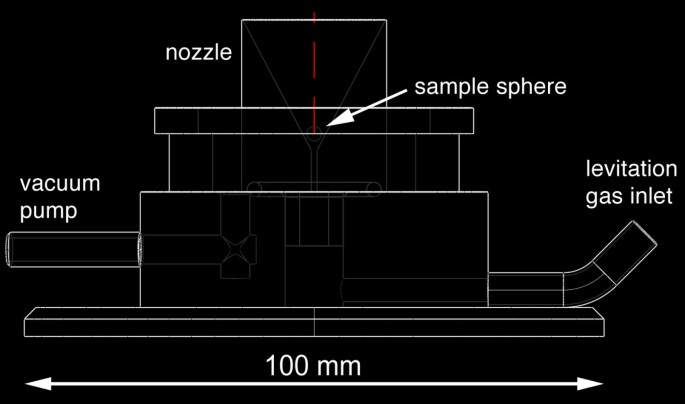
Cross department through the lower office of the levitation apparatus (sample chamber removed). The levitation nozzle comprised a 0.viii mm bore and an opening angle of 60°.
Aerodynamic levitation experiments required solid, nearly spherical samples as starting material. One way of preparing such a starting sample was to briefly fire the light amplification by stimulated emission of radiation (defocused to ~2 mm, ~1 southward) directly into a pot with the sample pulverisation. The pulverisation melted and solidified every bit nearly spherical burnished droplet. The glass beads could be placed upside down, i.due east. with the molten surface directed toward the levitation gas stream in the levitation nozzle. Alternatively, the sample powder (~ten - 20 mg) could be placed in a spectroscopy-grade graphite crucible (~five mm inner diameter, ~four mm depth) and briefly melted into a glass bead. The resultant bead could exist placed in the nozzle for levitation.
Stable levitation was a prerequisite for sample homogenization for chemical analyses too equally for evaporation and reduction experiments. The nozzle with the sixty° opening angle and 0.8 mm bore allowed to keep molten silicates in a stable position for up to >1 h (Fig. 4). We tested too a nozzle with a 0.8 mm bore, just a wider opening angle of 120°. However, we could not keep the spheres in stable positions with this nozzle. The gas period was set to 250 - 300 mL min-1 for silicate spheres with ~2 mm bore. Smaller as well as larger spheres failed to float stably. The distance between sphere and nozzle was ~ane mm. The aluminum metallic nozzle did not reach temperatures exceeding ~100°C even in the long-run experiments at full light amplification by stimulated emission of radiation power.
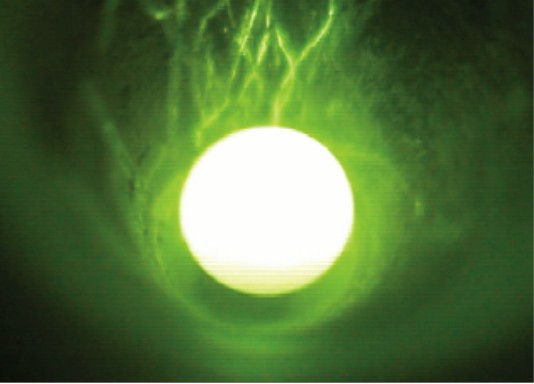
Video prototype showing a droplet of liquid basalt BCR-two during levitation. The sample was heated from the peak using a CO2-laser. The diameter of the sphere was ~ii mm.
Sample preparation for bulk rock chemical analyses
In order to demonstrate if aerodynamic levitation melting is suitable for major and minor element bulk rock analyses, we prepared glassy spheres of 7 United States Geological Survey (USGS) standard stone powders: BIR-1 (basalt, 48.0 wt.% SiO2), BHVO-ii (basalt, 49.9 wt.% SiO2), West-2 (diabase, 52.vii wt.% SiO2), BCR-two (basalt, 54.1 wt.% SiO2), AGV-2 (andesite, 59.iii wt.% SiO2), GSP-ii (granodiorite, 66.6 wt.% SiO2), and RGM-1 (rhyolite, 73.4 wt.% SiOii). Bulk compositions (recommended values) from the GeoReM database [38] were taken.
In training for levitation, the stone standard powders were pre-fused in modest graphite crucibles. A new crucible was machined for each sample in order to avoid any cross contamination. The pre-fused sample beads were placed upside downwards, i.e. with the burnished surface in the levitation nozzle, levitated and melted between 1 and 4 times for ~5 s each in an Ar temper (i.e. we used Ar as levitating gas). Argon gas was used because we wanted to avoid changes in oxidation country of the samples. High concentrations of ferric Fe in chondritic samples that were levitated in air led to formation of abundant <10 μm sized quenched spinel crystals [28–30].
The attached video system was used to monitor the fusion process. Remaining crystals were visible every bit bright spots in the slowly rotating and convecting melts. Only samples with no visible crystals left were used for the chemical analyses. The disappearance of crystals indicated that the liquidus temperature was reached. Samples were fused at temperatures only piffling above their liquidus in guild to minimize evaporation.
We quenched the samples by switching off the laser. The cooling rates for aerodynamic levitation experiments with one mm forsterite spheres were reported to be in the range of 700 °C southward-one [19]. As a result of the high cooling rates and absence of sites for heterogeneous nucleation, all silicate rock standards solidified as glass.
The spheres were removed from the nozzle by means of a pair of tweezers after they cooled downward in the Ar gas stream. They were stored in small drinking glass vials. We embedded the glass spheres into resin and prepared polished 1-inch sections. The sections were carbon coated before EPMA. One department contained up to >xl spheres.
We used a JEOL 8900R electron microprobe for major and minor element (Si, Ti, Al, Mg, Atomic number 26, Mn, Na, K; Kα lines) chemic analyses of the USGS rock standard glasses. Nosotros used a defocused beam (25 μm) with acceleration voltage of 15 kV and an electron current of 15 nA. The microprobe was calibrated with a set of natural and synthetic silicates and oxides. The scale was complemented by analyses of reference glasses KL2, GOR128, GOR132, ML3B, T1, StHs6/80 and ATHO [27].
We analyzed 10 randomly selected spots on each of the USGS rock standard glasses. The results of EPMA of the investigated 7 USGS stone standards are listed in Tabular array 1. The internal heterogeneity (major and pocket-size elements, EPMA spots) of the spheres was in all cases <5% (1σ SE, N = ten).
We have displayed all data with normalization on the concentration of the respective reference value of the element and on Al (Figs. five, 6, vii and 8). The measured to reference ratios were normalized to Al because this was the most refractory [i.e. has the highes evaporation temperature; [31, 39]] major element in the samples and was not expected to be lost past evaporation. This is mutual practice when displaying chemical data with respect to volatility. A ratio of one indicates that the respective chemical element was not lost during melting. A ratio <1 indicates evaporation. In such a case, the respective value gives the fraction of the element that remained in the sample. A ratio exceeding unity can only be due to sample heterogeneity.

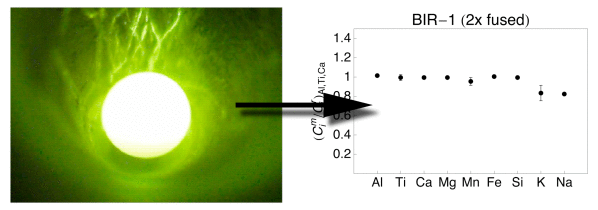
Plots showing the major and minor element composition of quenched spheres (1 - four times fused) of basaltic standards BIR-i and BHVO-2. The element concentrations were normalized to the reference concentrations [38] and to Al (meet text for details). The 1σ error bars are outlined.
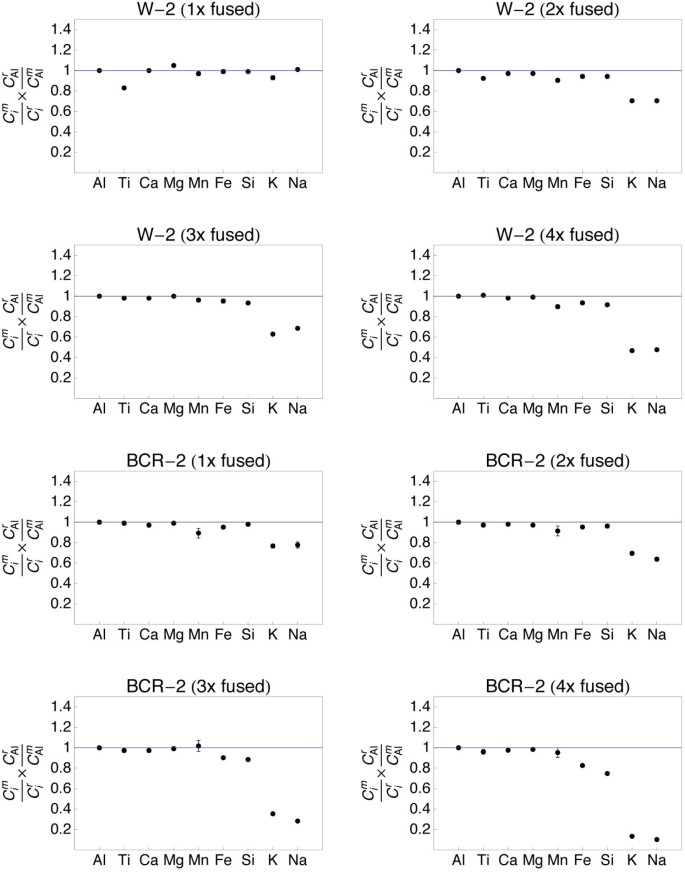
Plots showing the major and minor element limerick of quenched spheres (one - 4 times fused) of standards W-2 and BCR-2. The element concentrations were normalized to the reference concentrations [38] and to Al (see text for details). The 1σ error confined are outlined.
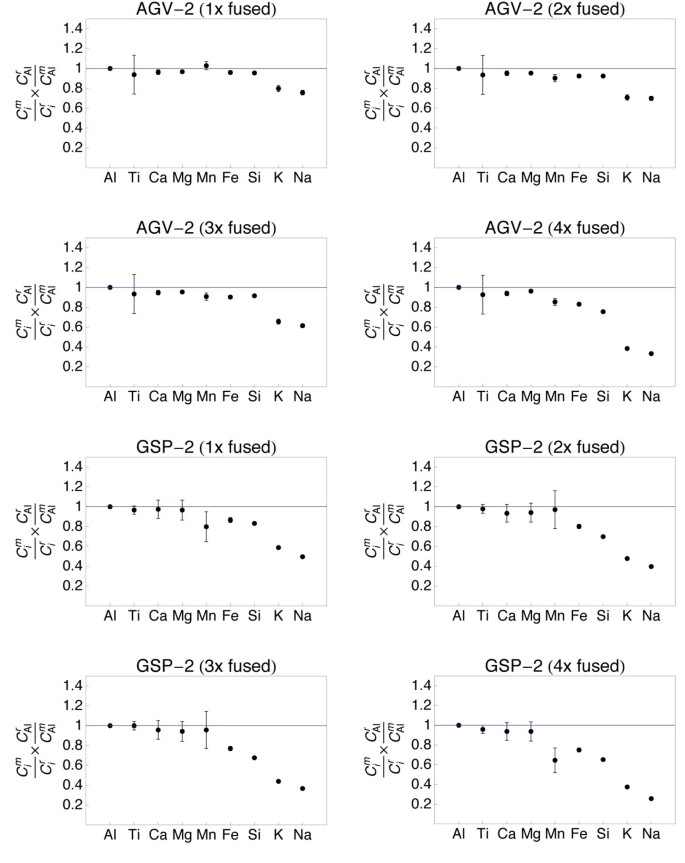
Plots showing the major and small element composition of quenched spheres (1 - 4 times fused) of andesite AGV-2 and granitoid GSP-2. The element concentrations were normalized to the reference concentrations [38] and to Al (meet text for details). The 1σ error bars are outlined.
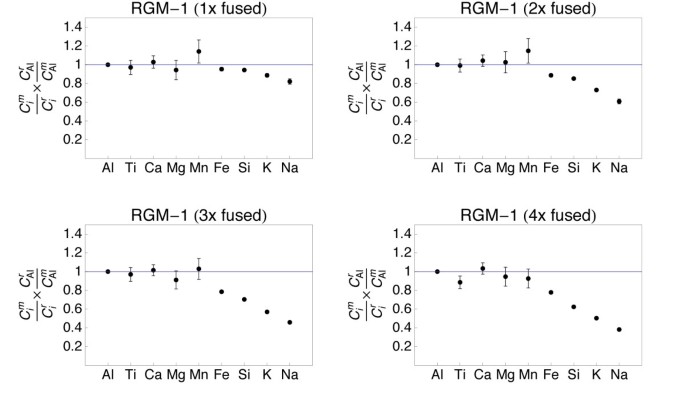
Plots showing the major and minor element composition of quenched spheres (1 - iv times fused) of rhyolite standard RGM-one. The chemical element concentrations were normalized to the reference concentrations [38] and to Al (see text for details). The 1σ mistake bars are outlined.
The refractory chemical element (Al, Ti, Ca, Mg) data from all samples demonstrate that homogeneity increases with the number of fusion steps. A unmarried fusion resulted in significant deviation of e.thou., Atomic number 26 (BIR-1, Fig. v) or Ti (W-2, Fig. 6) from the normalized reference concentrations. Subsequent fusion steps removed the heterogeneities. Increasing the number of fusion steps, even so, besides led to increasing loss of moderately volatile elements Na and Chiliad and, with more intense fusion, also Si and Fe (Figs. 5, 6, 7 and 8).
It is obvious from Figs 5, 6, seven and 8 that the moderately volatile elements Na and K were lost from nearly samples, even when samples are levitated and fused only once. Exceptions were mafic samples BHVO-two (Fig. 5) and W-ii (Fig. half dozen), which showed no loss of Na and K when fused one time. All samples that were fused between 2 and 4 times show a loss of 20 - 90% of Na and K.
Samples, which have been 2 - 4 times levitation melted showed concentrations of Al, Ti, Ca and Mg that agreed within ± 5% with the respective reference values. Manganese, Atomic number 26 and Si likewise agreed within ± v% with the reference values, if samples were non fused more 3 times. Four times of melting led to loss of alkalis, but as well of up to xl% of Si from the felsic sample RGM-one (Fig. eight). Loss of Mn, Fe and Si was less astringent in the SiO2-poor, mafic materials.
Loss of Na and K was clearly related to the number of fusion steps (Fig. ix). The loss of Grand correlated with the loss of Na in all, except the most SiO2-rich samples. In the nearly SiOii-rich samples GSP-2 and RGM-1 the relative loss of Na was systematically higher than the loss of Thou (Fig. 9).

Plot of Na (open circumvolve) and K (open square) concentrations in 7 USGS rock standards normalized to the information reported for these materials in [38]. The number of fusion events is indicated (i - iv times).
Alkali evaporation experiments
Alkali (Na, K) evaporation experiments were conducted with a picritic basalt PB-63. The chemical composition of PB-63 was previously determined past X-ray fluorescence (XRF, Göttingen, Table 2). The liquidus temperature was 1620 °C. The picrite was pre-fused to a sphere in a graphite crucible before it was placed in the levitation nozzle. We used the closed sample chamber with Ar as levitation gas. The outlet of the levitation appliance was direct connected to the source of an ICPMS in social club to monitor the limerick of the evaporated material (Fig. ten). The sample was heated and fused with variable laser energy. We monitored the stability of the sphere using the video system.
Sketch illustrating the set up for the evaporation experiment. The exhaust of the levitation bedroom was continued to the inlet of the ICPMS. Argon was used as levitation gas.
The intensities of 23Na, 24Mg, 27Al, 29Si, 39K, 43Ca, 55Mn and 57Fe were continuously monitored with using a PERKIN ELMER DRC II quadrupole mass spectrometer. Dwell times of the isotopes were matched to achieve approximately the aforementioned intensities to minimize counting errors peculiarly when determining element ratios.
The relative sensitivity factors (RSFi) for an isotope j of chemical element i were calculated according to:
(i)
The RSF values were adamant by analyzing the aerosol that was released from the NIST 610 standard glass during ultra violet (UV, λ = 193 nm) excimer laser ablation (Tabular array three). In social club to average out daily variations, we accept adopted the hateful RSF values that were obtained over a period of viii month. Since aerosol from the UV light amplification by stimulated emission of radiation ablation and from evaporation experiments volition not necessarily have the same backdrop, concentration data from this study have a larger intrinsic error. It was, yet, the purpose of this experiment to demonstrate a possible application of aerodynamic levitation rather than determining the exact limerick of the evaporated textile.
We used Na as internal standard because nosotros expected that the Na bespeak exceeded the signals of other elements in the evaporated aerosol. Sodium was the almost volatile of the investigated elements [39]. The relative 1σ standard errors of the mean RSF values ranged from 2 to 27% with an average relative 1σ standard error of ± ten%.
The concentration Ci of element i in the aerosol was calculated by means of:
(2)
Equation 2 shows that the conclusion of C i requires knowing the concentration of the internal standard (i.east. Na). This was, a priori, not known for the aerosol that forms from the evaporated material. Except for oxygen, however, we monitored all major and modest elements (Na, Mg, Al, Si, K, Ca, Mn, Fe) that were nowadays in the evaporated picrite PB-63 (Table 2) and assumed that the sum of the elements in the aerosol amounted 100 wt.%.
Combining Eq. 2 and Eq. 3 thus allowed decision of the composition of the aerosol without knowledge of the concentration of the internal standard element Na. This mode, we acquired quantitative analyses of the evaporated material from the picrite Atomic number 82-63 with high time resolution.
During the evaporation experiment, we heated the picrite PB-63 with laser energies between 20 and 50 W (Fig. 11). The sample was entirely molten during the whole experiment. The duration of the experiment was ~17 min. Evaporation started at 20 W laser free energy, but became pronounced at 30 W (Fig. eleven). The full signal increased with increasing laser free energy and hence temperature of the melt (Fig. eleven). The response time of the system with respect to an increase in laser free energy was ~10 s. The betoken exponentially decayed later on switching off the laser with a one-half live of ~50 s (Fig. eleven).
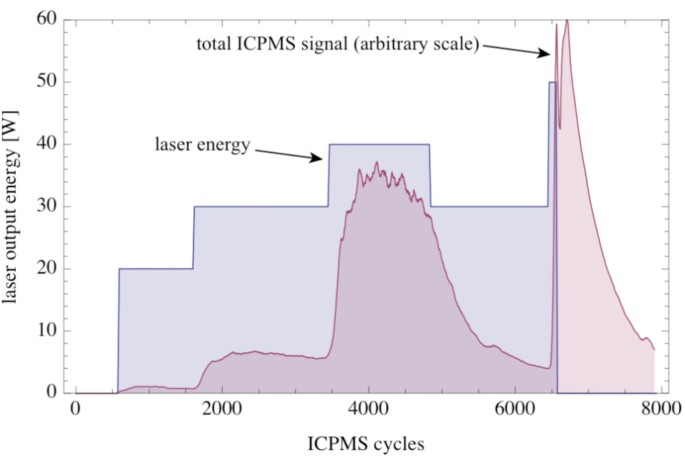
Plot of the laser output energy vs. the number of ICPMS cycles (7.seven cycls s -1 ; blueish). The crimson filled curve shows the normalized total ICPMS point (Na, Mg, Al, Si, K, Ca, Mn and Atomic number 26; arbitrary scale). The total ICPMS indicate was smoothed with a moving average of 20 cycles.
More than 80 wt.% of the aerosol was Na and Thousand (Fig. 12). Both elements showed a stiff correlation in the evaporated material. The Na/K-mass ratio in the evaporated material was in the range betwixt 0.9 and ane.ii. The amount of Si in the evaporated textile varied betwixt ~5 and ~18 wt.%. The Si-content increased with increasing temperature. Pocket-sized components in the evaporated fabric were Mg and Fe; both occurring in the 1 wt.% concentration level at the highest temperatures (Fig. xiii). Other elements were only present as trace components in the evaporated material.
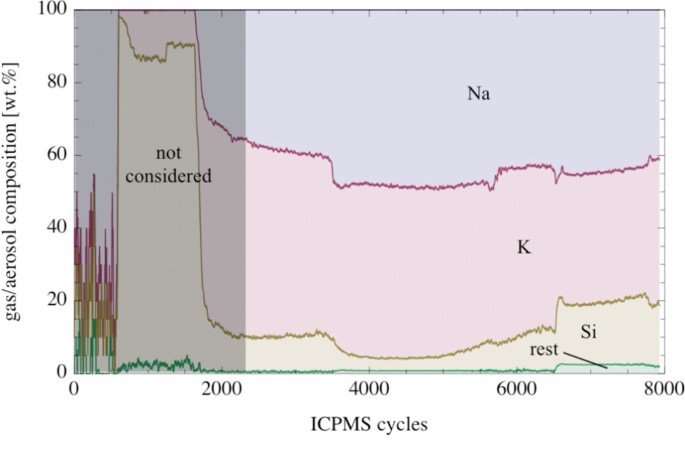
Plot of the composition of the evaporated gas/droplets from picrite Atomic number 82-63 vs. number of cycles (7.7 cycl due south -1 ). The signals were smoothed with a moving average of 20 cycles.
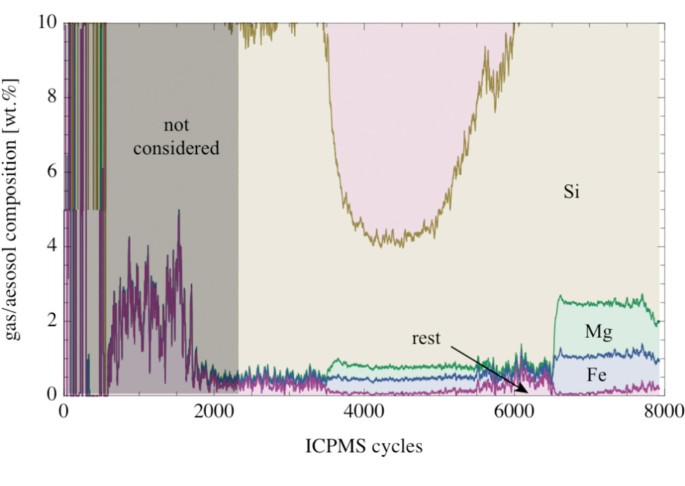
Plot of the composition of the evaporated material from picrite Atomic number 82-63 (enlarged part of Fig. 12). Small components in the evaporated phase are Mg and Fe (~ane wt.% of the oxide, respectively) with the remaining elements Al, Ca and Mn amounting <0.5 wt.%. The signals were smoothed with a moving boilerplate of 20 cycles.
Reduction experiments
The first reduction experiment was conducted with a mixture of natural spinel lherzolite powder (GZG1275/1, Table 2) that was mixed with 39 wt.% metal powder (88.vi wt.% Fe, 0.35 wt.% Co, 10.1 wt.% Ni). The metal mixture was prepared from analytical grade pure Iron, Co and Ni powders. The spinel lherzolite sample was provided by R. Schoenberg (Hannover) and was identical to the sample analyzed past Schoenberg et al., 2006 [40]. We have determined the chemical composition of the lherzolite by EPMA on a glassy sample that was homogenized by means of levitation melting (Table two), results are identical within doubtfulness. The liquidus temperature of the lherzolite was computed with MELTS [41, 42] at 1655°C. The lherzolite metal mixture was oxidized in a muffle furnace (~1000°C, ~3 h) in air before melting and levitation in social club to transfer the metal to oxides. The change in color of the powder from dark grey towards reddish brown suggested that metal was oxidized. The oxidized pulverization was re-footing and analyzed by ways of X-ray powder diffraction using a Phillips PW 1710 with Cu-Kα radiation (5° < 2Θ < lxx°, 0.02° steps, 0.5 s step-i). No Fe and Ni metal peaks were identified in the diffraction pattern. Instead, hematite (Atomic number 262O3) and bunsenite (NiO) and olivine were the predominant phases in the pattern.
The liquidus temperature of the oxidized lherzolite metallic mixture (see Table 2 for chemic limerick) was adamant with using MELTS [42] to be 1240°C with FeO-rich olivine as liquidus phase.
The starting material was fused in a graphite crucible to a pocket-size sphere. The sphere was placed in the nozzle in the closed sample chamber in social club to maintain a reducing atmosphere. The levitation gas was a commercially bachelor mixture of 98 vol.% Ar and 2 vol.% H2 for ~10 min. The oxide-lherzolite mixture was levitated and melted for about 5 min in the reducing gas atmosphere. The experimental run production consisted of a silicate fraction and metal (Fig. 14). The metallic sphere pointed towards the lesser of the levitation nozzle during melting. Both phases were liquid during the class of the experiment. The surface of the silicate sphere shows marks of skeletal olivine crystals that formed during quenching.

Photography of the reduced lherzolite-metal oxide mixture. Metal exsolved from the silicate. The diameter of the sphere was ~2 mm. The cloth was levitated with a mixture of 98 vol.% Ar and 2 vol.% Htwo.
For the 2nd reduction experiment, we used material of the NWA 869 L4-6 ordinary chondrite [43]. The chondrite powder was oxidized in air for ~24 h in order to transfer all metal into oxides. The sample was re-ground to powder in a paw mortar afterwards oxidation. No metal grains were left. The sample was briefly pre-fused in a graphite crucible in air. The glass bead was and so transferred into the levitation nozzle, where it was levitated using the 98 vol.% Ar + 2 vol% Htwo gas mixture. The sample was fused for about 30 s in the reducing atmosphere and quenched by switching off the laser. It was embedded into resin for electron back scattered electron imaging and EPMA.
The run product consisted of quenched olivine crystals, interstitial glass and round metal droplets (Fig. 15). The composition of the silicate portion was determined by x randomly selected EPMA spots (10 μm spot diameter, Table 4). The FeO/SiO2 mass ratio of the silicate portion was 0.67. The NiO/SiO2 ratio of the silicate portion was 0.004. The metal blebs were rich in Ni with Ni contents between 29.five and 59.8 wt.%. The concentration of Co was negatively correlated with Ni and varied betwixt 0.69 and 1.38 wt.%. The concentrations of Si were <0.05 wt.% in all metal blebs. The mean metal composition is listed in Table 4.

Dorsum scattered electron epitome of NWA 869 L4-6 ordinary chondrite. The sample was entirely oxidized prior to levitation melting. A 98 vol.% Ar + two vol.% H2 mixture was used equally levitating gas.
Discussion
Bulk rock chemical analyses
The sample preparation protocol was time efficient (~5 min per sample) and required merely 10 mg of sample powder. Testing different melting durations (Figs. 5, 6, 7 and eight) showed that 2 - iii brief fusion steps (~5 southward each) were the best compromise between sample homogeneity and loss of elements through evaporation. Non-volatile major and minor elements Si, Ti, Al, Ca, Mn and Fe could be adamant with an doubt <v% relative (Tabular array two). Moderately volatile elements Na and K could not exist adamant with observable accuracy considering they evaporate during sample melting (Fig. 9). The results demonstrated that levitation melting was a suitable preparation technique for bulk refractory elements, but not suited for moderately volatile elements like Na and K. The results on major and small not-volatile elements support the conclusion by Pack et al. [28], Pack [29] and Patzer et al. [30] that aerodynamic levitation melting is a suitable method for the grooming of bulk rocks for analyses of refractory elements.
Alkali evaporation experiments
The test with picrite Lead-63 demonstrated that aerodynamic levitation in combination with ICPMS is a promising new technique for the investigation of evaporation from silicate melts. The experiment showed that Na and Thousand evaporated in the same relative proportions from the cook. The Na/K mass ratio in the evaporated material (0.95 - one.two) is like to the ratio in the picrite PB-63 (Na/Thousand = ane.22, Table 2). This observation was in agreement with the observed evaporative loss of Na and K from the USGS rock standards; except for the nearly silica rich samples (Fig. 9). This suggests that Na and K were similarly volatile nether experimental atmospheric condition (1 bar, Ar-atmosphere). The nigh SiO2-rich rock samples (GSP-ii, RGM-1) showed a preferred evaporation of Na relative to One thousand (Fig. nine). The divergence is likely related to a higher ratio of the alkali oxide activity coefficients of felsic melts relative to mafic melts. An increase in γ leads to an increase in vapor pressure and hence elevated evaporation rates.
The response fourth dimension to changes in laser energy were in the range of tens of seconds and allowed studies of evaporation processes with high time-resolution. Chondrules formed within a few minutes [34] in the solar nebula. Therefore, aerodynamic levitation in combination with ICPMS is a suitable technique to monitor evaporation of alkalis under different conditions (melt composition, temper, temperatures). A smaller sample chamber may reduce the response times.
Reduction experiments
The exam with the lherzolite-oxide mixture demonstrated that siderophile elements could be extracted from silicate by means of levitation melting in reducing atmosphere. Siderophile trace elements can be extracted along with Atomic number 26 and Ni by sample reduction. We demonstrated (Fig. 14) that levitation in an Ar-H2 mixture allows reduction of a major fraction of Fe and Ni to metallic. Highly siderophile elements (e.g., platinum grouping elements) are expected to be quantitatively concentrated in the metal. Laser ablation ICPMS analyses of siderophile trace elements accept been successfully conducted on fe meteorites [44, 45].
The reduction experiment with the oxidized chondrite material showed that reduction of oxides (FeO, Fe2O3, NiO) to metal alloys occurred within 30 s. The high Ni concentration in the resultant metal was due to the low caste of reduction. Nickel is more than siderophile than Co, which is more siderophile than Iron. It follows from the difference in redox potential that Ni is reduced first, followed past Co. The superchondritic Ni/Fe (0.85) and Co/Iron ratios (0.021) in the metallic in the run production are clearly the issue of the preferential reduction of Ni and Co. The chondritic ratios are 0.058 (Ni/Iron) and 0.0028 (Co/Atomic number 26) [39]. The Fe/Si ratio of the silicate is only fiddling below the L-chondritic ratio. This demonstrates that most Fe remained in the silicate. The low Ni/Atomic number 26 ratio in the silicate is bear witness that most Ni was reduced to metal.
Chondrule formation took place in a more than reduced surround of the solar nebula, which was essentially H2, and chondrule melting lasted over the range of minutes to tens of minutes. It is therefore plausible that metal in chondrites has formed past reduction during the brief chondrule melting event [36].
Conclusions
Aerodynamic levitation in combination with microchemcial methods (EPMA, LA-ICPMS) is an efficient means of bulk rock preparation for analyses of not-volatile major, minor and trace elements. The strength of the technique lies in the absence of contamination and the option to analyze just small (~x mg) samples. A further reward is that no flux is used, which would dilute the elements of interest and could crusade contagion, namely for trace and ultra trace elements. A disadvantage of the sample preparation technique involving containerless fusion is evaporative loss of moderately volatile elements like Na and K.
Aerodynamic levitation in combination with ICPMS online monitoring of the evaporated material allows time-resolved studies of the evaporation behavior at high temperatures. It is demonstrated that Na and One thousand are similarly volatile at ane bar Ar atmosphere. This observation is confirmed by results from fused rock standards.
Levitation and melting experiments in reduced atmosphere demonstrated that metallic and silicate could be separated past reduction. The exsolution of metallic did not impact the stability of the levitated melt. The experiment with an oxidized chondrite showed that Ni-rich metal is exsolved within ~30 south of melting.
References
-
Sandhage KH, Yurek GJ: Indirect dissolution of sapphire into silicate melts. Periodical of the American Ceramic Order. 1988, 71: 478-489. 10.1111/j.1151-2916.1988.tb05898.x.
-
Malvin DJ: Silica-glass containers for high-temperature experiments. American Mineralogist. 1988, 73: 1198-1200.
-
Grove T: Use of FePt alloys to eliminate the iron loss problem in 1 atmosphere gas mixing experiments: Theoretical and practical considerations. Contributions to Mineralogy and Petrology. 1982, 78: 298-304. x.1007/BF00398924.
-
Johannes Due west, Bode B: Loss of iron to the Pt-container in melting experiments with basalts and a method to reduce information technology. Contributions to Mineralogy and Petrology. 1978, 67: 221-225. x.1007/BF01046578.
-
Nordine PC, Atkins RM: Aerodynamic levitation of laser-heated solids in gas jets. Review of Scientific Instruments. 1982, 53: 1456-1464. ten.1063/i.1137196.
-
Oran WA, Berge LH: Containerless melting and solidificating of materials with an aerodynamic levitation arrangement. Review of Scientific Instruments. 1982, 53: 851-853. 10.1063/1.1137067.
-
Ouaida MB, Badie JM: Dispositif de levitation aérodynamique associe à un concentrateur solaire pour I'étude de materiaux liquides. Journal of Physics (Series E: Scientific Instrumentation). 1982, 15: 941-944. x.1088/0022-3735/15/ix/021.
-
Landron C, Hennet L, Thiaudiere D, Price DL, Greaves GN: Structure of liquid oxides at very loftier temperatures. Nuclear Instruments and Methods in Physics Research. 2003, B 199: 481-488.
-
Weber JKR, Benmore C, Tangeman JA, Siewenie J, Heira KJ: Structure of binary CaO-AliiO3 and SrO-Al2O3 liquids by combined levitation-neutron diffraction. The Journal of Neutron Research. 2002, 11: 113-121. 10.1080/1023816031000099746.
-
Glorieux B, Millot F, Rifflet J-C: Surface tension of liquid alumina from contactless techniques. International Periodical of Thermophysics. 2002, 23: 1249-1257. 10.1023/A:1019848405502.
-
Glorieux B, Millot F, Rifflet J-C, Coutures J-P: Density of superheated and undercooled liquid alumina past a conactless method. International Periodical of Thermophysics. 1999, 20: 1085-1094. 10.1023/A:1022650703233.
-
Landron C, Hennet L, Thiaudiere D: Combining measurements of synchrotron radiation and neutrons from liquids at loftier temperature. Belittling Sciences. 2001, 17: 165-168.
-
Nordine PC, Weber JKR, Abadie JG: Properties of loftier-temperature melts using levitation. Pure and Applied Chemical science. 2000, 72: 2127-2136. 10.1351/pac200072112127.
-
Arai Y, Aoyama T, Yoda Due south: Spherical sapphire single-crystal synthesis by aerodynamic levitation with high growth rate. Review of Scientific Instruments. 2004, 75: 2262-2265. 10.1063/ane.1763254.
-
Blander M, Pelton Ad, Jung I-H, Weber R: Non-equilibrium concepts lead to a unified explanation of the formation of chondrules and chondrites. Meteoritics and Planetary Science. 2004, 39: 1897-1910. ten.1111/j.1945-5100.2004.tb00085.x.
-
Nagashima K, Moriuchi Y, Tsukamoto K, Tanaka Thousand, Kobatake H: Critical cooling rates for glass formation in levitated Mg2SiO4-MgSiOthree chondrule melts. Journal of Mineralogical and Petrological Sciences. 2008, 103: 204-208. x.2465/jmps.070620c.
-
Nagashima K, Tsukamoto K, Satoh H, Kobatake H, Dold P: Reproduction of chondrules from levitated, hypercooled melts. Journal of Crystal Growth. 2006, 293: 193-197. 10.1016/j.jcrysgro.2006.01.064.
-
Weber JKR, Tangeman JA, Cardinal TS, Heira KJ, Paradis P-F, Ishikawa T, Yu J, Yoda S: Novel synthesis of calcium oxide-aluminum oxide glasses. Japanese Journal of Applied Physica. 2002, 41: 3029-3030. 10.1143/JJAP.41.3029.
-
Tangeman JA, Phillips BL, Navrotsky A, Weber JKR, Hixson Advertising, Primal TS: Vitreous forsterite (Mg2SiO4): synthesis, construction and thermochemistry. Geophysical Enquiry Messages. 2001, 28: 2517-2521. 10.1029/2000GL012222.
-
Skinner LB, Barnes AC, Crichton Due west: Novel behaviour and structure of new glasses of the type Ba-Al-O and Ba-Al-Ti-O produced by aerodynamic levitation and light amplification by stimulated emission of radiation heating. Journal of Physics: Condensed Affair. 2006, 18: L407-10.1088/0953-8984/18/32/L01.
-
Albarède F: Geochemistry: an introduction. 2003, Cambridge University Press, two
-
Eggins S: Laser ablation ICP-MS analysis of geological materials prepared as lithium borate glasses. Geostandards and Geoanalytical Research. 2003, 27: 147-162. 10.1111/j.1751-908X.2003.tb00642.ten.
-
Nehring F, Jacob D, Barth M, Foley S: Laser-ablation ICP-MS analysis of siliceous stone glasses fused on an iridium strip heater using MgO dilution. Microchimica Acta. 2008, 160: 153-163. ten.1007/s00604-007-0819-vii.
-
Stoll B, Jochum KP, Herwig Thousand, Amini M, Flanz M, Kreuzburg B, Kuzmin D, Willibold Yard, Enzeweiler J: An automatic iridium-strip heater for LA-ICPMS bulk analysis of geological samples. Geostandards and Geoanalytical Research. 2007, 32: v-26.
-
Yu Z, Norman M, Robinson P: Major and trace element assay of silicate rocks by XRF and laser ablation ICP-MS using lithium borate fused glasses: matrix effects, instrument response and results for international reference materials. Geostandards and Geoanalytical Research. 2003, 27: 67-89. 10.1111/j.1751-908X.2003.tb00713.10.
-
Sylvester PJ: Trends in belittling developments and earth science applications in LA-ICP-MS and LA-MC-ICP-MS for 2004 and 2005. Geostandards and Geoanalytical Research. 2006, thirty: 197-207. 10.1111/j.1751-908X.2006.tb01062.x.
-
Jochum KP, Stoll B, Herwig K, Willbold M, Hofmann AW, Amini M, Aarburg S, Abouchami W, Hellebrand E, Mocek B, et al: MPI-DING reference glasses for in-situ microanalysis: new reference values for element concentrations and isotope ratios. Geochemistry Geophysics Geosystems. 2006, vii: Q02008-ten.1029/2005GC001060.
-
Kropf A, Pack A: Primordial alkalis in chondrules. Geochimica et Cosmochimica Acta. 2007, 71: A526-10.1016/j.gca.2006.09.026.
-
Pack A: Fractionation of refractory lithophile elements in bulk chondrites and chondrite components. Lunar and Planetary Scientific discipline Conference. 2008
-
Patzer A, Pack A, Gerdes A: Zirconium and hafnium in meteorites. Meteoritics and Planetary Science. 2010
-
Palme H: Chemical and isotopic heterogeneity in protosolar matter. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences. 2001, 359: 2061-2075. 10.1098/rsta.2001.0897.
-
Alexander C, Grossman JN, Ebel DS, Ciesla FJ: The formation conditions of chondrules and chondrites. Scientific discipline. 2008, 320: 1617-1619. 10.1126/science.1156561.
-
Borisov A, Pack A, Kropf A, Palme H: Partitioning of Na between olivine and cook: An experimental written report with application to the formation of meteoritic NatwoO-rich chondrule glass and refractory forsterite grains. Geochimica et Cosmochimica Acta. 2008, 72: 5558-5573. ten.1016/j.gca.2008.08.009.
-
Zanda B: Chondrules. Earth and Planetary Science Letters. 2004, 224: 1-17. 10.1016/j.epsl.2004.05.005.
-
Galy A, Young ED, Ash RD, Keith O'Nions R: The formation of chondrules at loftier gas pressures in the Solar Nebula. Science. 2000, 290: 1751-1753. x.1126/scientific discipline.290.5497.1751.
-
Connolly HC, Huss GR, Wasserburg GJ: On the germination of Iron-Ni metallic in Renazzo-similar carbonaceous chondrites. Geochimica et Cosmochimica Acta. 2001, 65: 4567-4588. 10.1016/S0016-7037(01)00749-ix.
-
Pack A, Yurimoto H, Palme H: Petrographic and oxygen-isotope study of refractory forsterites from R-chondrite Dar al Gani 013 (R3.5-vi), unequilibrated ordinary and carbonaceous chondrites. Geochimica et Cosmochimica Acta. 2004, 68: 1135-1157. x.1016/j.gca.2003.07.014.
-
Jochum KP, Nohl U, Herwig K, Lammel E, Stoll B, Hoffmann AE: GeoReM: a new geochemical database for reference materials and isotopic standards. Geostandards and Geoanalytical Inquiry. 2005, 29: 333-338. ten.1111/j.1751-908X.2005.tb00904.x.
-
Lodders K: Solar system abundances and condensation temperatures of the elements. The Astrophysical Periodical. 2003, 591: 1220-1247. 10.1086/375492.
-
Schoenberg R, von Blanckenburg F: Modes of planetary-scale Fe fractionation. World and Planetary Scientific discipline Letters. 2006, 252: 342-359. x.1016/j.epsl.2006.09.045.
-
Ghiorso MS, Sack RO: Chemic mass transfer in magmatic processes. IV. A revised and internally consequent thermodynamic model for the interpolation and extrapolation of liquid-solid equilibria in magmatic systems at elevated temperatures and pressures. Contributions to Mineralogy and Petrology. 1995, 119: 197-212. x.1007/BF00307281.
-
Asimov PD, Ghiorso MS: Algorithmic modifications extending MELTS to summate subsolidus phase relations. American Mineralogist. 1998, 83: 1127-1131.
-
Conolly HC, Zipfel J, Grossman JN, Folco L, Smith C, Jones RH, Righter Thou, Zolensky M, Russell SS, Bendix GK, et al: The meteoritical bulletin, No. 90, 2006 September. Meteoritics and Planetary Science. 2006, 41: 1383-1418. 10.1111/j.1945-5100.2006.tb00529.x.
-
Campbell Air conditioning, Humayun M: Trace element microanalysis in iron meteorites by laser ablation ICPMS. Analytical Chemical science. 1999, 71: 939-946. 10.1021/ac9808425.
-
Mullane E, Alard O, Gounelle M, Russell SS: Laser ablation ICP-MS study of IIIAB irons and pallasites: constraints on the behaviour of highly siderophile elements during and after planetesimal core formation. Chemical Geology. 2004, 208: 5-28. 10.1016/j.chemgeo.2004.04.024.
Acknowledgements
A. Thomen (Nancy, France) is thanked for his aid in early stages of the evolution of the levitation apparatus. The work of Chiliad. Tondock, who congenital the levitation apparatus (nozzle, sample chamber) is appreciated. A. Kropf wrote a helpful LABVIEW program that allowed graphical control of the gas flow. Y. Xiao is thanked for providing the stone standards. Corrections by A. Patzer improved the English.
Effective comments past the AE and an anonymous reviewer helped to improve the manuscript.
Author data
Affiliations
Corresponding author
Additional data
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
AP designed the levitation device and conducted first experiments. AP wrote the manuscript. KK and AK conducted the EPMA analyses. NA prepared the USGS samples. KS, NA and AP conducted the evaporation experiments and ICPMS analyses of the evaporated materials. All authors accept read and approved the final manuscript.
Authors' original submitted files for images
Rights and permissions
Open Access This commodity is distributed nether the terms of the Creative Commons Attribution ii.0 International License (https://creativecommons.org/licenses/by/two.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reprints and Permissions
Nearly this article
Cite this commodity
Pack, A., Kremer, K., Albrecht, North. et al. Clarification of an aerodynamic levitation appliance with applications in World sciences. Geochem Trans 11, 4 (2010). https://doi.org/ten.1186/1467-4866-xi-4
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/1467-4866-11-4
Keywords
- Instrumental Neutron Activation Analysis
- United States Geological Survey
- Majority Stone
- Solar Nebula
- Volatile Element
Source: https://link.springer.com/article/10.1186/1467-4866-11-4
0 Response to "A Review of Some Approximate Methods Used in Aerodynamic Heating Analyses"
Post a Comment